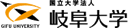RESEARCH
Research
Biosynthesis and functions of N-glycan branches
Overview of N-glycan branched structures
We work on the expression, functions, and disease-involvement of N-glycans attached to proteins. Particularly, we focus on the branched structures of glycans.
N-glycosylation is a fundamental protein modification with large structural variations. A key feature is its 2 to 5 branched structures. Each branch has its own function and is linked to specific diseases (Fig. 1). Moreover, formation of each branch is protein-selective and occurs only on a limited set of proteins. However, it is still poorly understood at the molecular level how each branch functions and how such protein selectivity is accomplished.
We tackle these questions by focusing on the biosynthetic enzymes (glycosyltransferases). We expect that our findings will ultimately lead to the development of new therapeutics for glycan-related diseases.
Bisecting GlcNAc, an accelerator of Alzheimer's disease
(Reference: Kizuka et al., EMBO Mol. Med., 2015)
(Reference: Nakano et al., Mol. Cell. Proteomics, 2019)
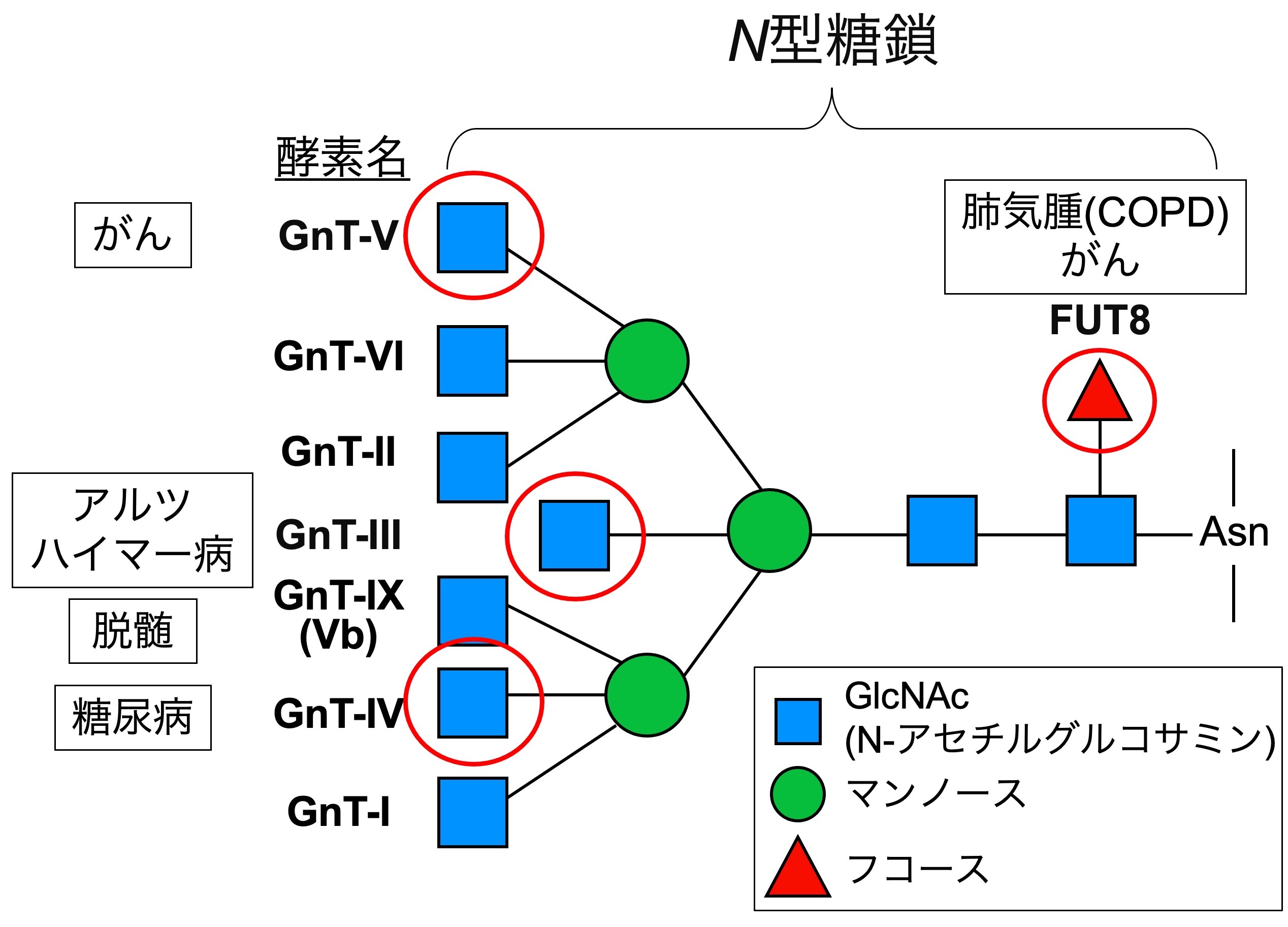
Bisecting GlcNAc is one of the N-glycan branches (Fig. 2, upper). We have revealed that this glycan structure is profoundly involved in the development and progression of Alzheimer's disease (AD). AD is known to be caused by deposition of amyloid-β (Aβ) peptide in the brain, and we found that bisecting GlcNAc-deficient mice show reduced Aβ deposition and improved AD pathology (Fig. 2, lower). Mechanistically, the Aβ-producing enzyme BACE1 is modified with bisecting GlcNAc, and BACE1 function is positively regulated by this glycan modification. Furthermore, bisecting GlcNAc levels are increased in AD patients, suggesting its involvement in AD progression in humans. However, we still do not fully understand how BACE1 function is regulated by bisecting GlcNAc at a molecular level. We now examine this mechanism and analyze the structure of the biosynthetic enzyme of bisecting GlcNAc, MGAT3 (GnT-III).
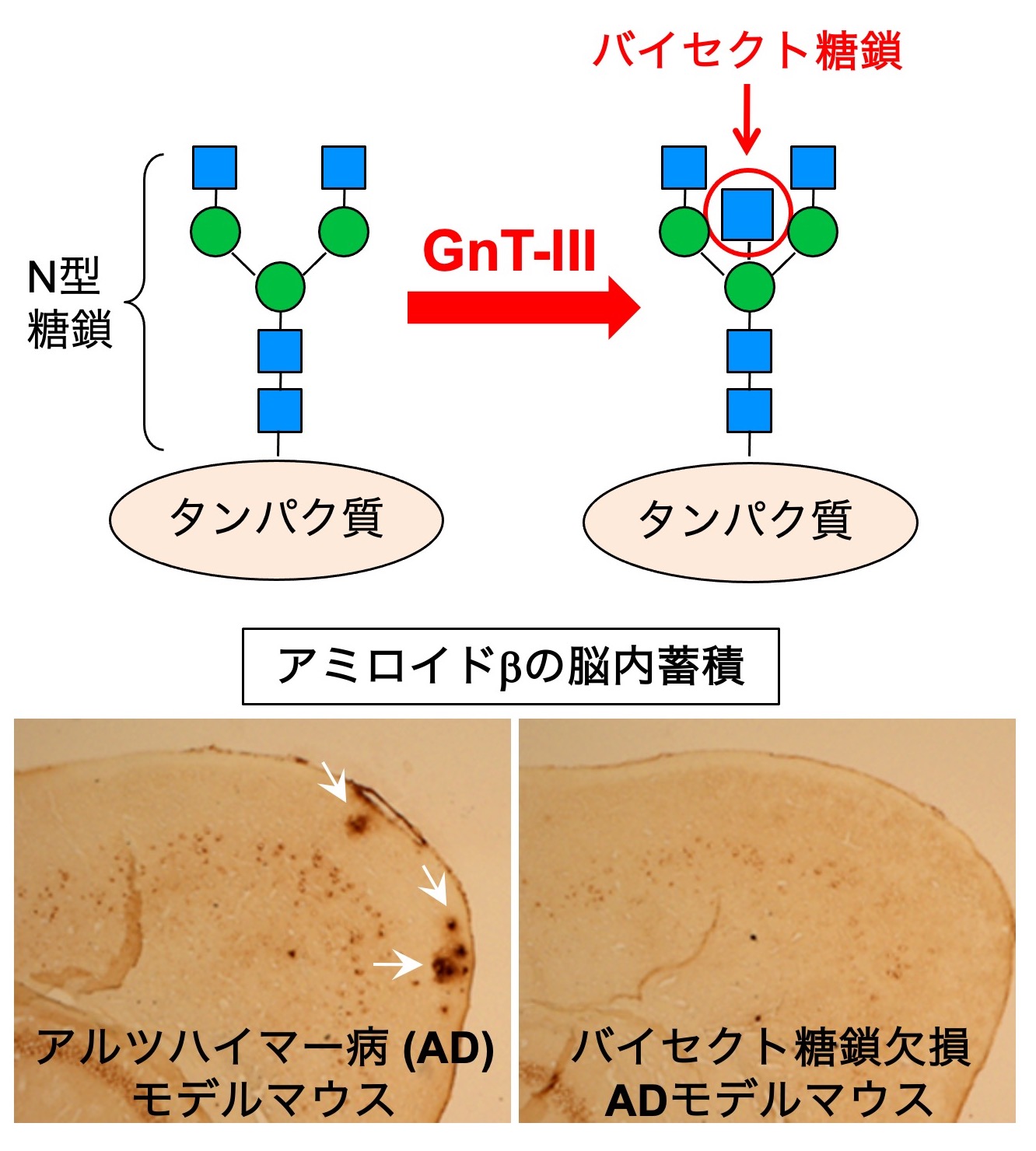
We also examine the intrinsic physiological functions of bisecting GlcNAc. By looking at the 3D structure of bisecting GlcNAc-containing glycans (Fig. 3), we found that the upper branch in N-glycans points in the opposite direction in the presence of bisecting GlcNAc, compared with N-glycans without bisecting GlcNAc. This results in suppression of further extension and branching of N-glycans, which normally occurs in non-bisected glycans. Therefore, bisecting GlcNAc has a suppressive role for N-glycan biosynthesis.
We now investigate the functions of bisecting GlcNAc in neurons and kidney, where bisecting GlcNAc is highly expressed, and we recently found that bisecting GlcNAc plays an important role in maintaining body fluid balance in the kidney. We also examine the regulatory mechanisms of MGAT3.
β1,6-branch, an accelerator of cancer
(Reference: Nagae et al., Nat. Commun., 2018)
(Reference: Osuka et al., J. Biol. Chem., 2022)
(Reference: Osuka et al., iScience, 2025)
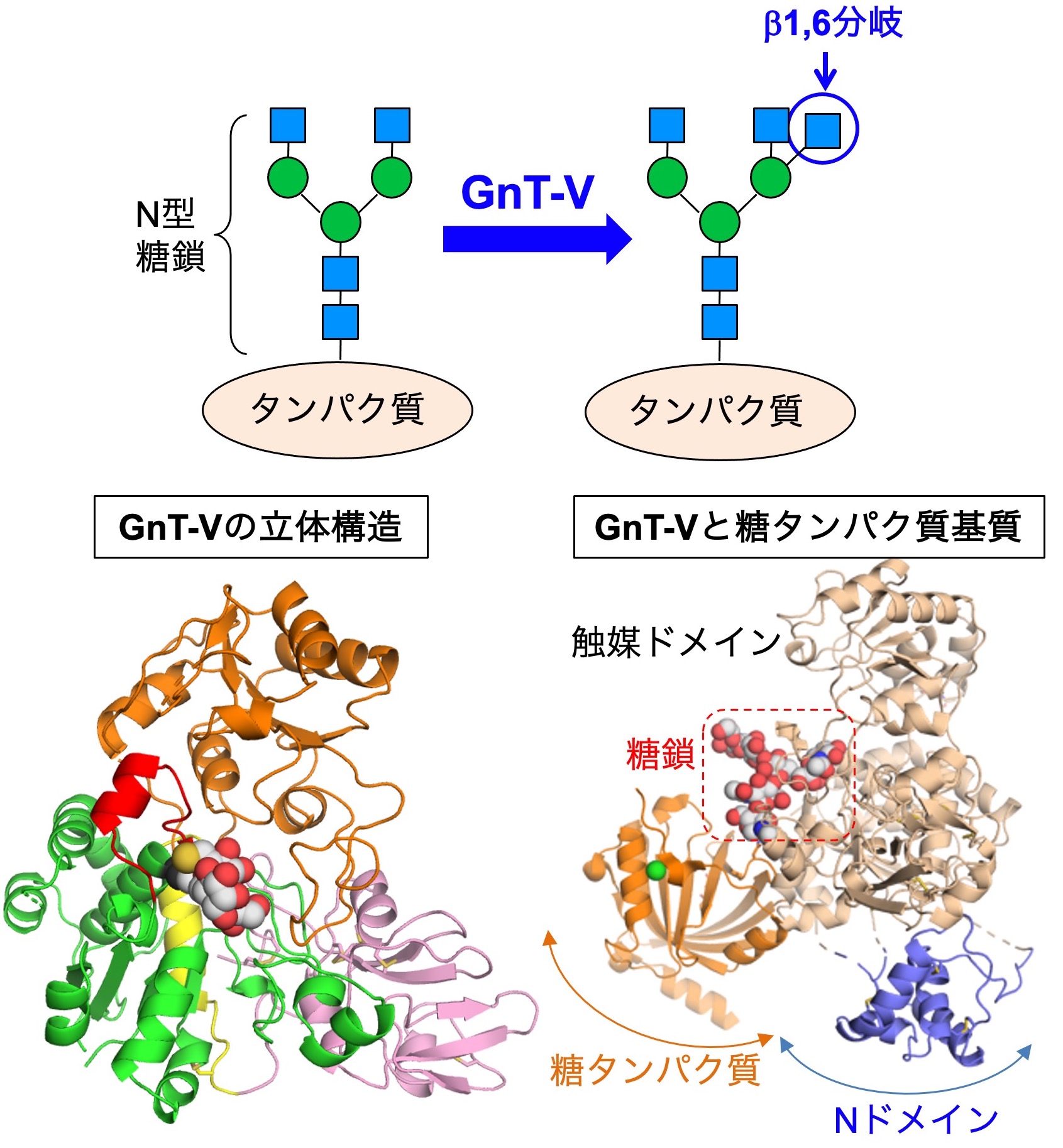
We also focus on N-glycan branches that are highly involved in cancer. Particularly, the β1,6-branch (Fig. 4, upper) synthesized by the MGAT5 (GnT-V) enzyme is known to promote cancer growth and metastasis. Therefore, it has been considered a promising drug target for many years.
We were the first to reveal the 3D crystal structures of MGAT5 in collaboration with an expert in structural biology (Dr. Masamichi Nagae) (Fig. 4, lower left). This provided basic information regarding how MGAT5 recognizes substrate glycans and biosynthesizes the β1,6-branch. The clarification of the structure will allow us to design specific MGAT5 inhibitors. Indeed, in collaboration with organic chemists, we are trying to develop MGAT5 inhibitors.
However, the detailed mechanisms of how MGAT5 selects target glycoproteins and works in living cells remain poorly understood. To address this question, we recently found that the N domain of MGAT5 (Fig. 4, lower right) is important. A mutant MGAT5 lacking the N domain almost completely lacks activity toward glycoproteins, while it almost fully retains activity toward glycans. This suggests that MGAT5 recognizes its substrate proteins through the N domain.
Recently, to investigate how MGAT5 selects proteins for glycosylation, we examined the mouse kidney to identify the proteins modified by MGAT5. We found that two proteins, ANPEP and Meprinα, located on the apical side of the proximal tubules, are the major substrate proteins of MGAT5 (Fig. 5). Our findings indicate that, in the mouse kidney, whether MGAT5-mediated glycosylation occurs is primarily determined by the transport of proteins to the apical side and by the three-dimensional structures of the substrate proteins. We are currently working on the mechanisms by which MGAT5 function is regulated in cells.
β1,4-branch related to diabetes
(Reference: Nagae et al., Commun. Biol., 2022)
(Reference: Osada et al., J. Biol. Chem., 2022)
(Reference: Osada et al., iScience, 2024)
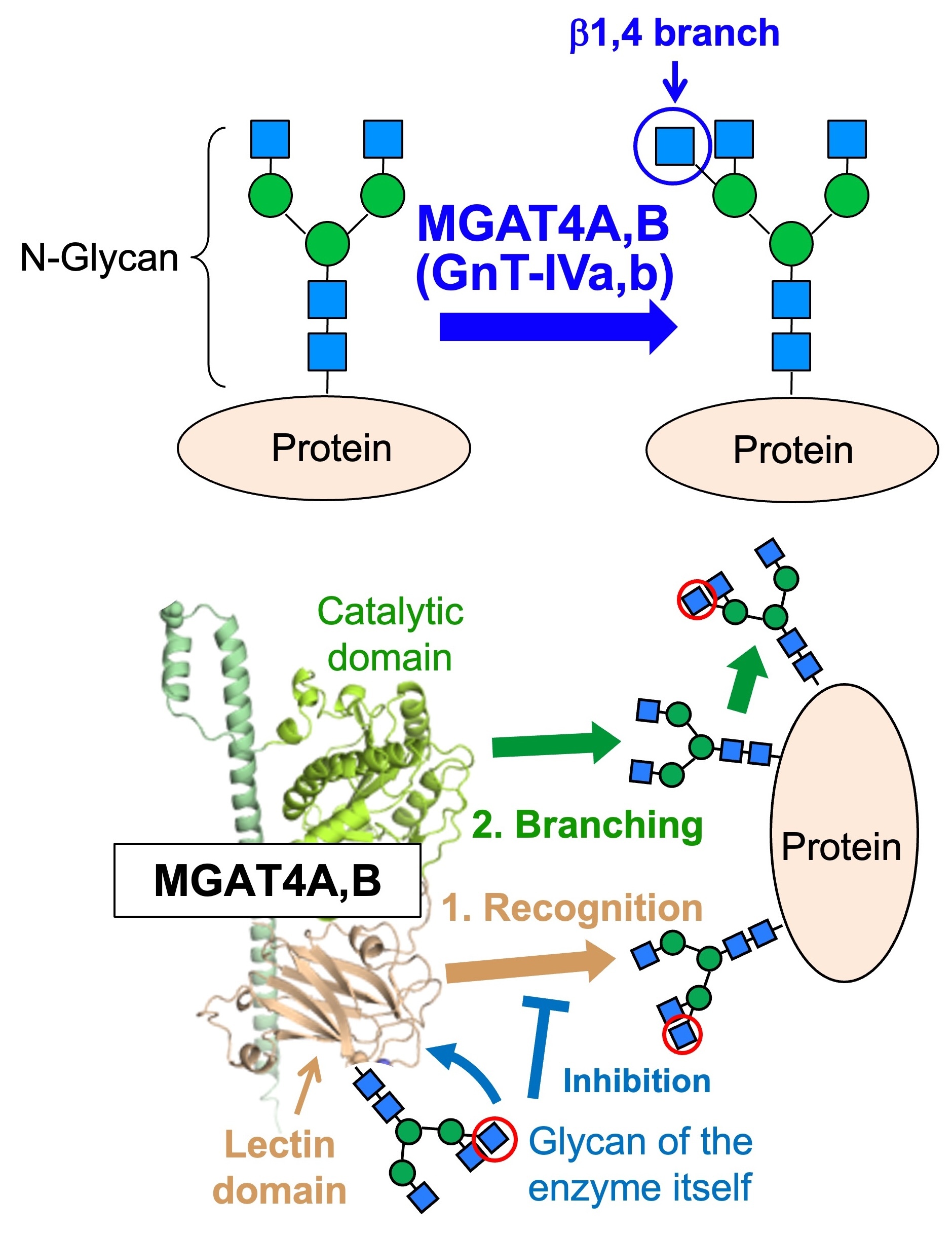
Another topic is the β1,4-branch, which is related to diabetes. This branch is biosynthesized by either of two similar enzymes, MGAT4A (GnT-IVa) or MGAT4B (GnT-IVb) (Fig. 6, upper). Because MGAT4A-deficient mice show impaired pancreatic function, leading to diabetic phenotypes with high blood glucose levels, this branch is expected to be a drug target for diabetes. On the other hand, it remains largely unclear how MGAT4A and 4B synthesize glycans in cells, and how the functions of these two similar enzymes differ.
We recently found that MGAT4A and 4B have a unique lectin domain not present in other related glycosyltransferases. Moreover, the lectin domain binds to specific glycans, and this binding is required for the enzyme activity of the catalytic domain (Fig. 6, lower). Furthermore, MGAT4A and MGAT4B possess their own glycans within the lectin domain. Since these glycans inhibit the function of the lectin domain in a glycan structure-dependent manner, MGAT4 self-regulates its activity through its own glycan. No other N-glycan biosynthetic enzyme has been shown to possess such a self-regulatory mechanism, suggesting that MGAT4A and MGAT4B synthesize glycans in a novel manner.
We also recently revealed that the MGAT4 family has not only A and B but also C through G members, and that the number of these genes and their enzymatic activity vary widely among species. Thus, MGAT4 plays a crucial role in synthesizing species-specific N-glycan branches.
Core fucose, a versatile sugar structure relevant to COPD and cancer
(Reference: Tomida et al., J. Biol. Chem., 2020)
(Reference: Tomida et al., J. Biol. Chem., 2022)
(Reference: Tomida et al., J. Biol. Chem., 2026)
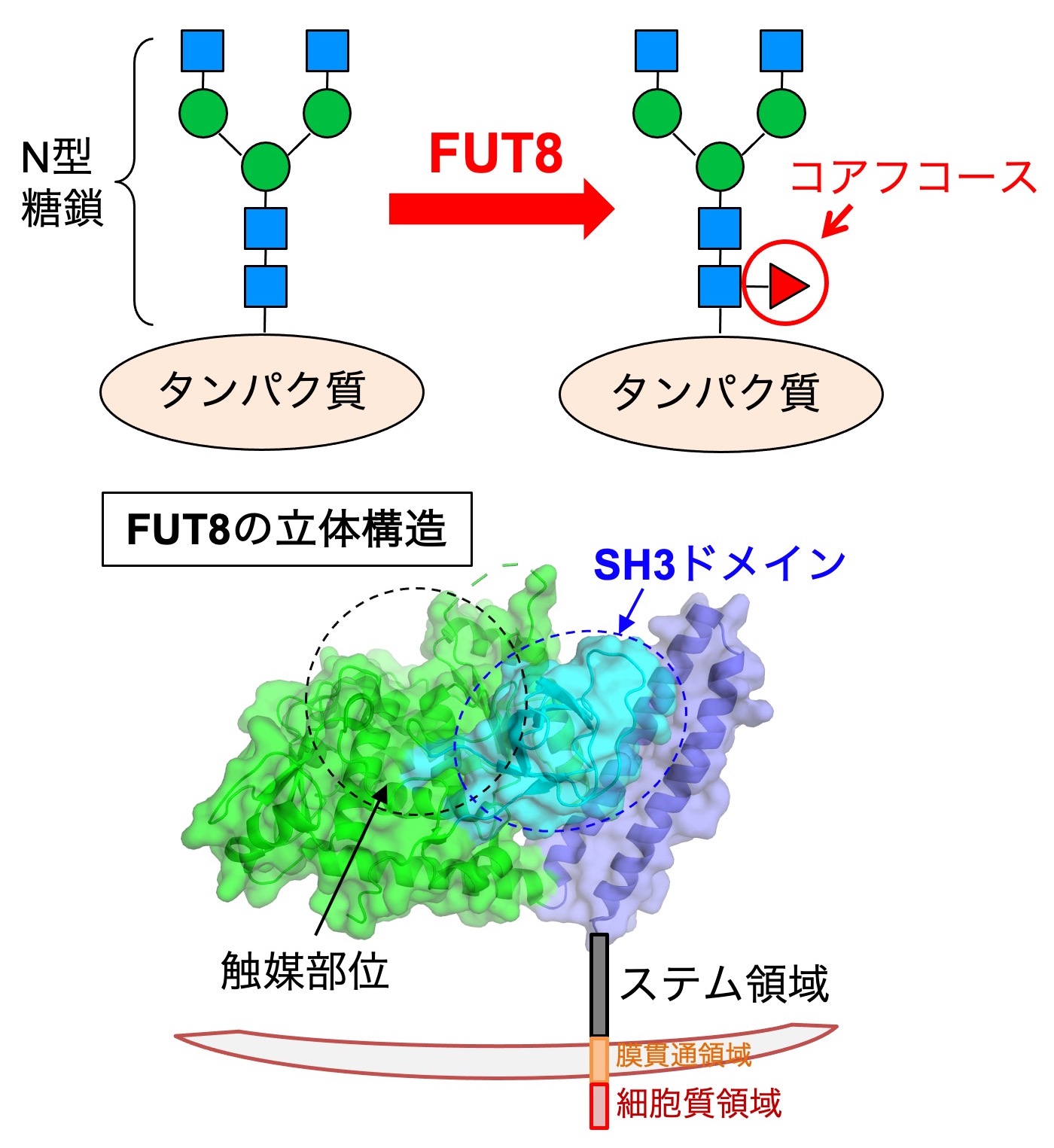
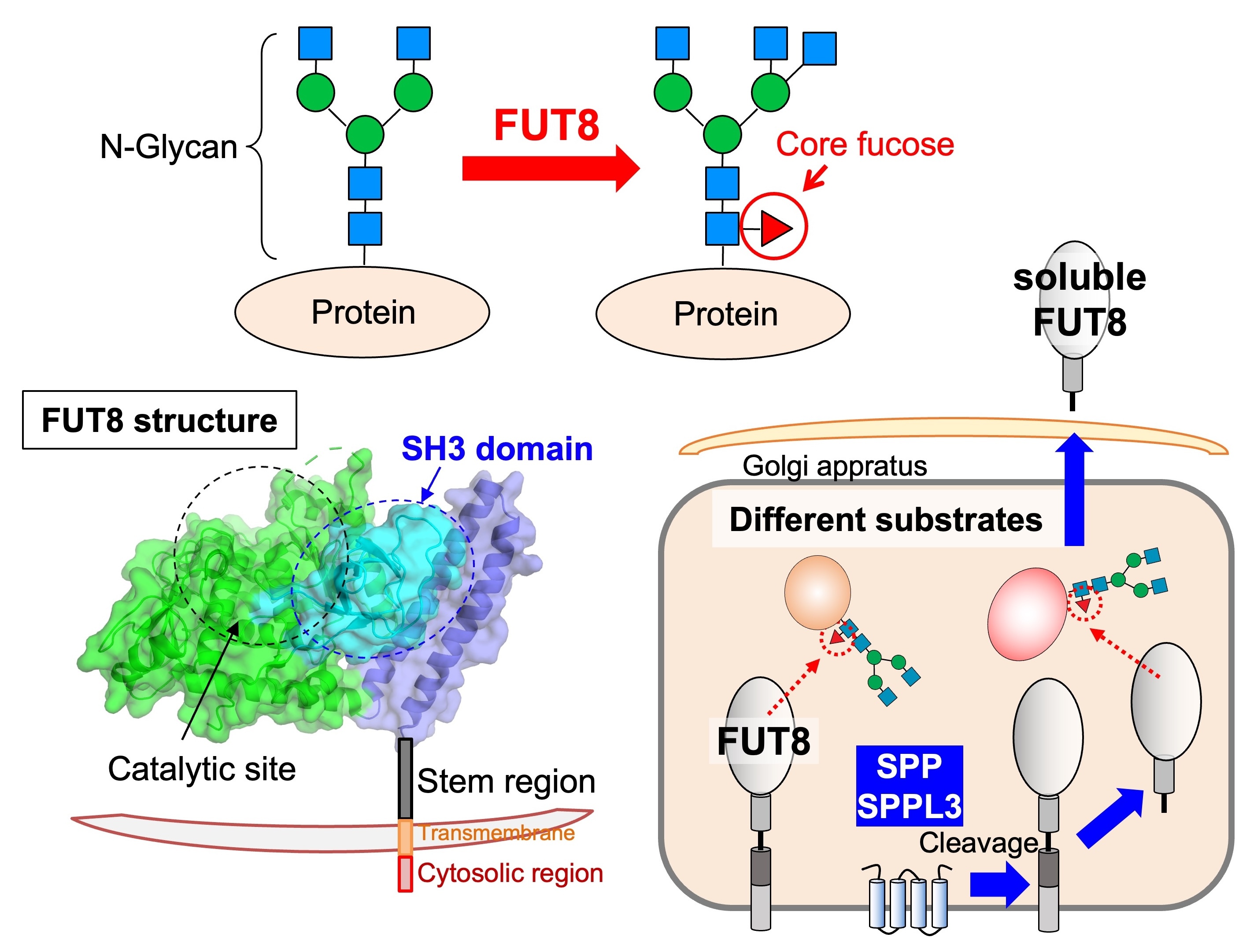
We also work on "core fucose", a fucose branch on the core part of N-glycans. Core fucose is biosynthesized by an enzyme called FUT8 (Fig. 7, upper) and is known to be involved in chronic obstructive pulmonary disease (COPD), as FUT8-deficient mice show an emphysema phenotype. In addition, core fucose has been reported to promote lung cancer and melanoma. Furthermore, a core-fucose-targeted technique is clinically used in which the anti-tumor activity of antibody drugs is dramatically increased by removing core fucose from the antibody. Therefore, core fucose is highly relevant to the diagnosis and therapy of these diseases.
The regulatory mechanisms of FUT8 functions remain to be clarified. We examine how FUT8 functions are regulated in cells by focusing on its 3D structure and regulatory proteins. The 3D structure of FUT8 is already known, and it has a unique SH3 domain near the catalytic domain (Fig. 7, lower left). We recently found that the SH3 domain is necessary for the enzyme activity of FUT8 and for interaction with the RPN1 protein. We also clarified that the stem region of FUT8 is required for its oligomerization.
In addition, we discovered that FUT8 is cleaved by SPP and SPPL3 proteases and secreted (Fig. 7, lower right). This cleavage affects the substrate protein selectivity of FUT8, indicating that core fucosylation activity of FUT8 in cells is regulated by its cleavage and secretion.
Projects on other glycosyltransferases
O-mannose glycans associated with brain function
(Reference: Itoh et al., J. Biol. Chem., 2026)
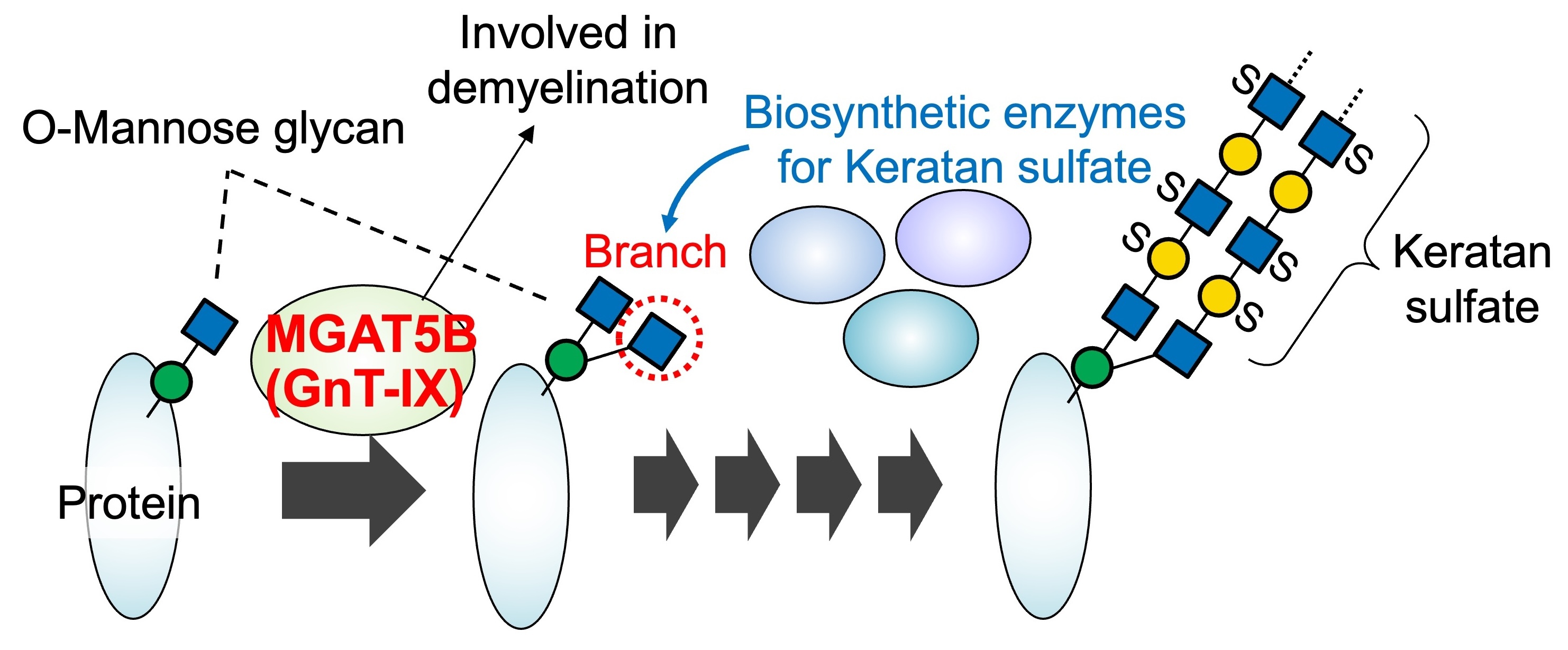
We work on a brain-specific glycosyltransferase, MGAT5B (GnT-IX), which is associated with brain function and disorders. MGAT5B synthesizes a branch in O-mannose glycans and has been shown to be involved in demyelinating diseases and brain tumors. We demonstrated that the MGAT5B-produced branch is recognized by other glycosylation enzymes, leading to the efficient formation of a glycan structure called keratan sulfate on the branched O-mannose glycans (Fig. 8). Currently, we are analyzing MGAT5B-deficient mice to investigate the physiological functions of branched O-mannose glycans in the brain.
LacdiNAc, a regulator of protein stability in blood and cancer
(Reference: Tokoro et al., J. Biol. Chem., 2024)
(Reference: Tokoro et al., J. Biol. Chem., 2026)
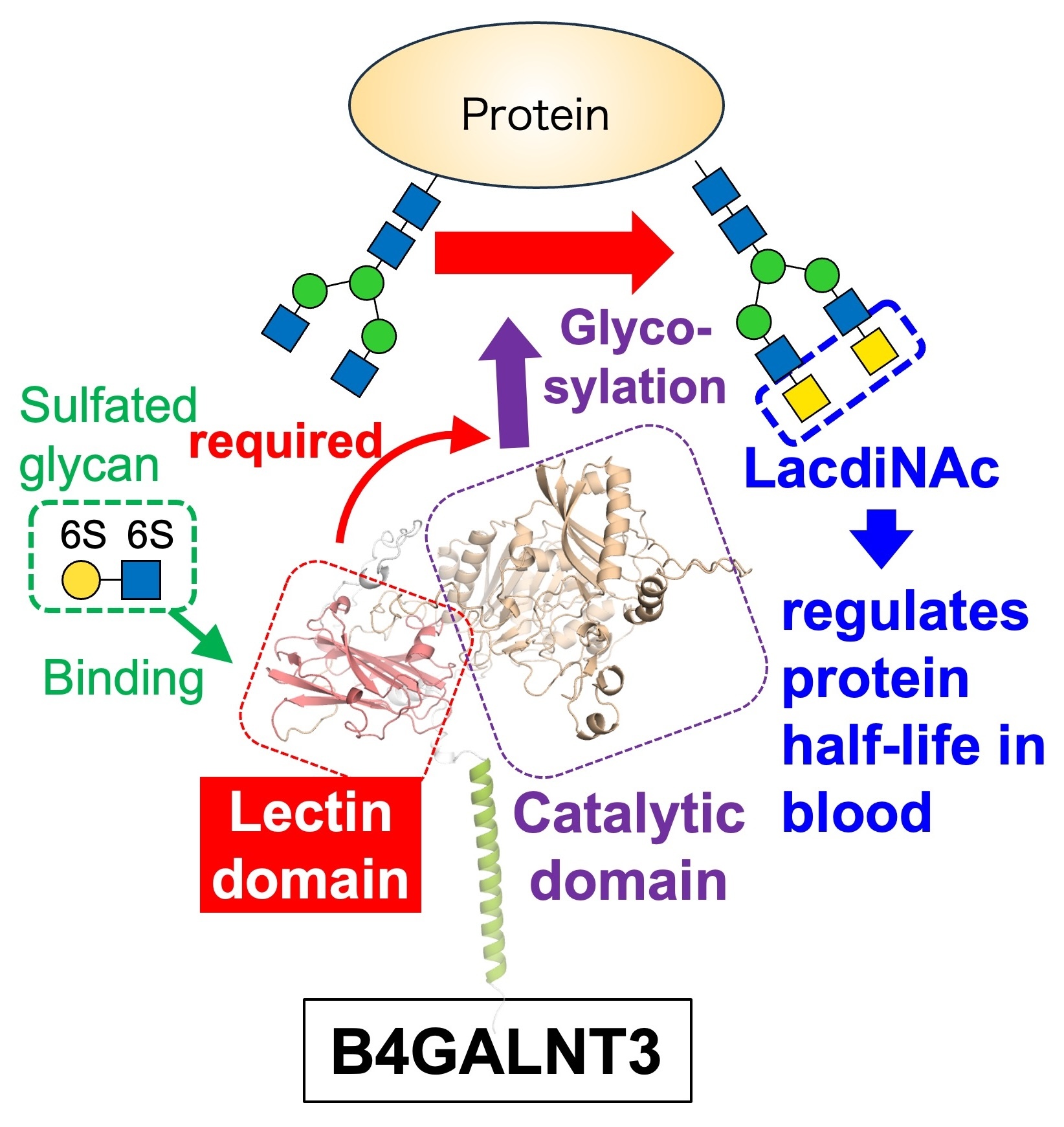
We investigate the unique structure and functions of a glycosyltransferase, B4GALNT3. It synthesizes a glycan designated LacdiNAc, which regulates the stability of proteins in the blood. Structural analysis of B4GALNT3 revealed that, in addition to a catalytic domain, it possesses a lectin domain that recognizes certain glycan structures. We found that this lectin domain is necessary for the action of B4GALNT3 toward glycoproteins, and that it binds to sulfated glycans (Fig. 9).
Furthermore, we found that once the LacdiNAc structure is formed by B4GALNT3, the biosynthesis of terminal structures such as sialic acid and HNK-1 is suppressed. This indicates that B4GALNT3 functions to inhibit glycan maturation and simplify glycan structures. We are currently examining the possible association of B4GALNT3 with several diseases.
Regulatory mechanisms of glycosyltransferase activity in cells
We also work on various mechanisms by which glycosyltransferase activity is regulated in cells. In more detail, we focus on subcellular localization, post-translational modifications, complex formation, degradation, and secretion of glycosyltransferases.
Comprehensive analysis of activity of all human glycosyltransferases (HGA)
Our group participates in the Human Glycome Atlas Project (HGA), one of the national projects funded by MEXT's Program for Promoting Large-scale Academic Frontier Projects. In this project, we are collecting all human glycosyltransferases and acquiring data on their enzymatic activities as foundational information for constructing a glycan biosynthesis atlas. By combining the obtained enzyme activity data with other information within the project, we aim to improve the accuracy of simulators that predict glycan expression from gene expression in cells.
Other research projects
New glycan probes and inhibitors
(Reference: Kizuka et al., Cell Chem. Biol., 2016)
(Reference: Kizuka et al., Cell Chem. Biol., 2017)
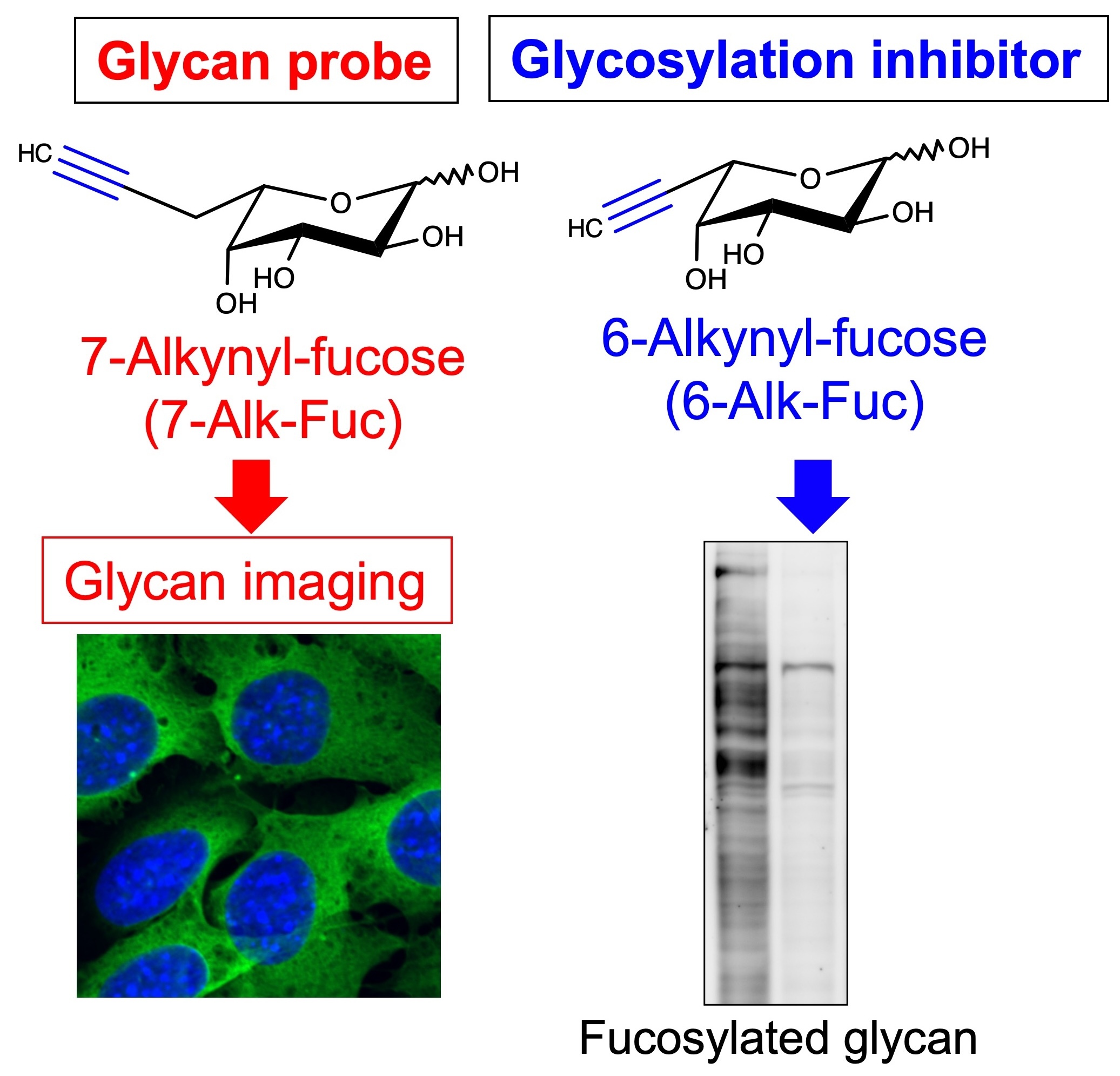
It is essential for any glycobiology study to detect the glycan of interest. Although common detection methods use antibodies or lectins, we focus on a different method using a combination of sugar analogs and click chemistry (Fig. 10, left). We aim to develop new drug targets and disease biomarkers using these new probes. In addition, for therapeutic purposes, we develop inhibitory compounds for glycan functions. So far, we have developed a fucosylation inhibitor that suppresses cancer cell invasion (Fig. 10, right). Furthermore, in collaboration with Dr. Hidenori Tanaka at Gifu University, we developed inhibitor candidates for MGAT5 (Vibhute et al., BBA Gen. Subj., 2022) and an inhibitor for glycosaminoglycan biosynthesis.
Small extracellular vesicles (sEVs) and glycans
This is a collaborative project with the Kenichi Suzuki Lab (iGCORE) on the expression and functions of glycans in small extracellular vesicles (sEVs), including exosomes. sEVs are drawing attention from many researchers because they function in cell-cell communication. We have demonstrated that a cancer-associated glycosyltransferase, MGAT5, is present in sEVs derived from cancer cells and is taken up by recipient cells (Reference: Hirata et al., iScience, 2023). Furthermore, we found that HNK-1, a brain-specific glycan, and its biosynthetic enzyme B3GAT1 (GlcAT-P) are also present in sEVs, and that HNK-1 glycan is similarly transferred between cells via sEVs (Reference: Tokoro et al., J. Biol. Chem., 2026).
GPI (Glycosyl Phosphatidyl Inositol) anchor
We work on the biosynthesis and functions of GPI, a unique glycolipid attached to proteins, particularly focusing on prion and mental diseases (Reference: Hirata et al., J. Biol. Chem., 2022). We also work on intracellular trafficking of GPI-anchored proteins.
Arginine methylation and glycans
This is a collaborative project with Assistant Prof. Misuzu Hashimoto (Faculty of Applied Biological Sciences, Gifu University). We work on the relationship between glycans/glycoproteins and arginine methylation, which is also known as a post-translational modification of proteins (Reference: Hashimoto et al., BBA Gen. Subj., 2020).
Biosynthetic regulation of HNK-1 (Human Natural Killer-1) glycan
We study the regulatory mechanisms of the HNK-1 glycan, which is specifically expressed in the brain and required for learning and memory functions. We have found that MGAT3 suppresses HNK-1 biosynthesis in a tissue-dependent manner (Reference: Kawade et al., Molecules, 2021).